

Autism Spectrum Disorder (ASD) represents one of the most complex challenges in modern pediatric neurology, characterized by a heterogeneous constellation of neurodevelopmental deficits that impact social communication, sensory processing, and behavioral regulation. Over the past few decades, the global prevalence of ASD has seen a staggering increase, with current data from the Centers for Disease Control and Prevention (CDC) suggesting that 1 in every 36 children in the United States is diagnosed with autism.
For many families, conventional pharmacological and behavioral interventions like Applied Behavior Analysis (ABA) provide a foundation for managing symptoms but often fail to address the underlying biological causes. This has led the scientific community and proactive parents toward exploring regenerative medicine. Specifically, the debate between Cord Blood Stem Cells and Autologous Stem Cells (from bone marrow or fat) has become a focal point for those seeking to modulate the neuroinflammatory pathways of autism.
This comprehensive guide provides an expert analysis of these two modalities, comparing their clinical efficacy, procedural nuances, and strategic benefits to help you make an informed decision for your child’s future.
Before choosing a stem cell source, it is vital to understand the scientific rationale behind their use. The transition of cellular therapy from an experimental modality to clinical application is rooted in the "immune-brain axis" hypothesis.
Research indicates that many children with ASD exist in a state of chronic neuroinflammation. In these children, the brain’s resident immune cells (microglia) adopt a pro-inflammatory M1 phenotype, leading to a “cytokine storm” (elevated IL-6 and TNF-α), which disrupts synaptic pruning and neural connectivity.
Both cord blood and autologous stem cell therapies aim to shift the brain from this pro-inflammatory state to an anti-inflammatory M2 state, promoting neurogenesis (the creation of new neurons) and improving cerebral perfusion, which helps restore function to regions of the brain responsible for social and language processing.
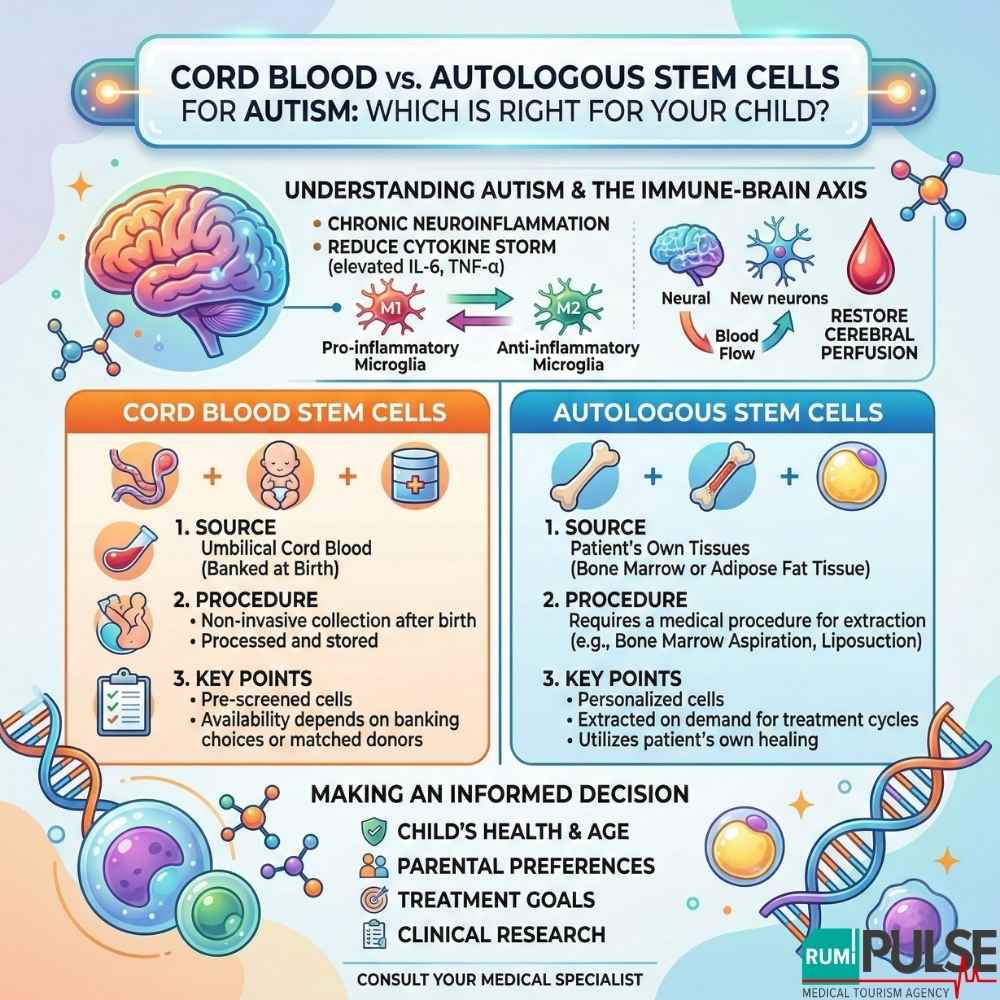
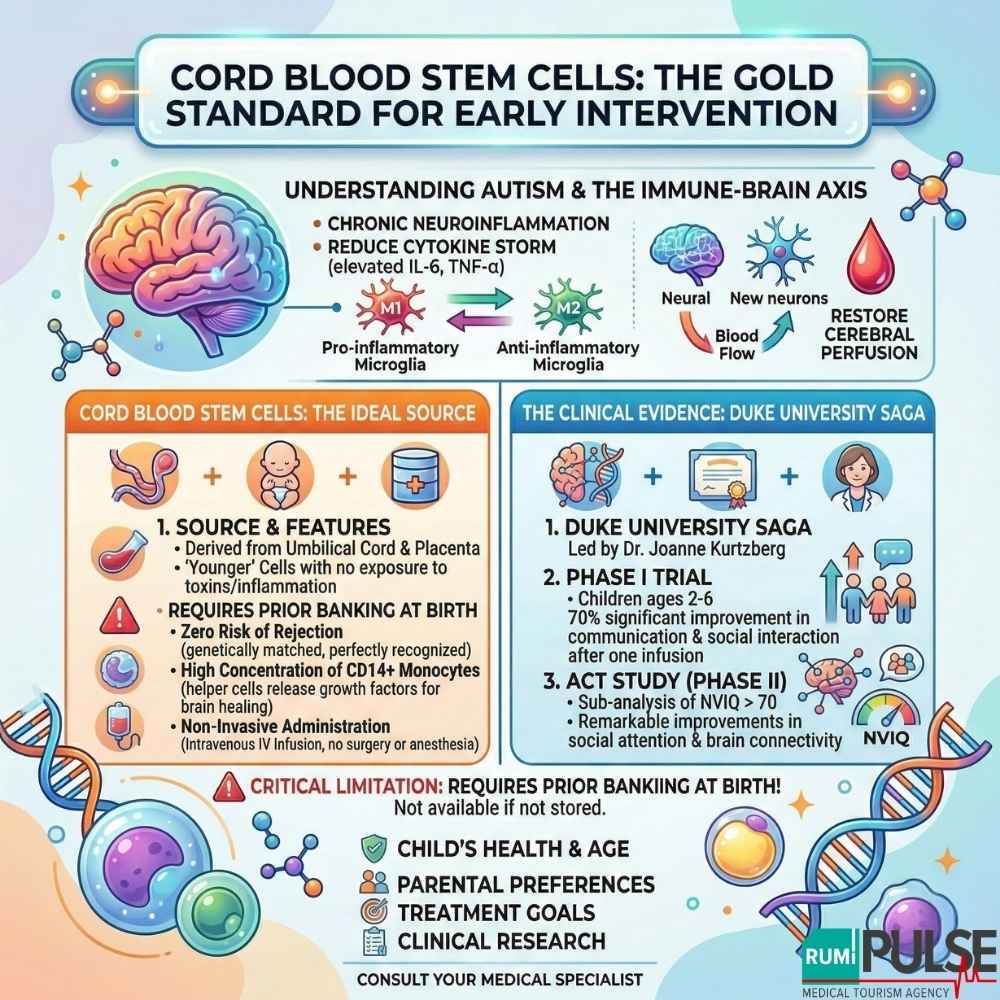
Cord blood stem cells are derived from the blood left in the umbilical cord and placenta after childbirth. These stem cells are unique because they are “younger” and have not been exposed to environmental toxins or the child’s internal inflammatory environment for years.
The most significant data comes from the research program at Duke University, led by Dr. Joanne Kurtzberg.
However, the primary limitation of cord blood therapy is availability. It requires prior banking at birth, meaning if you did not store your child’s cord blood, this specific option is unavailable.
For more information on cord blood stem cells and autism treatment, visit our contact page.
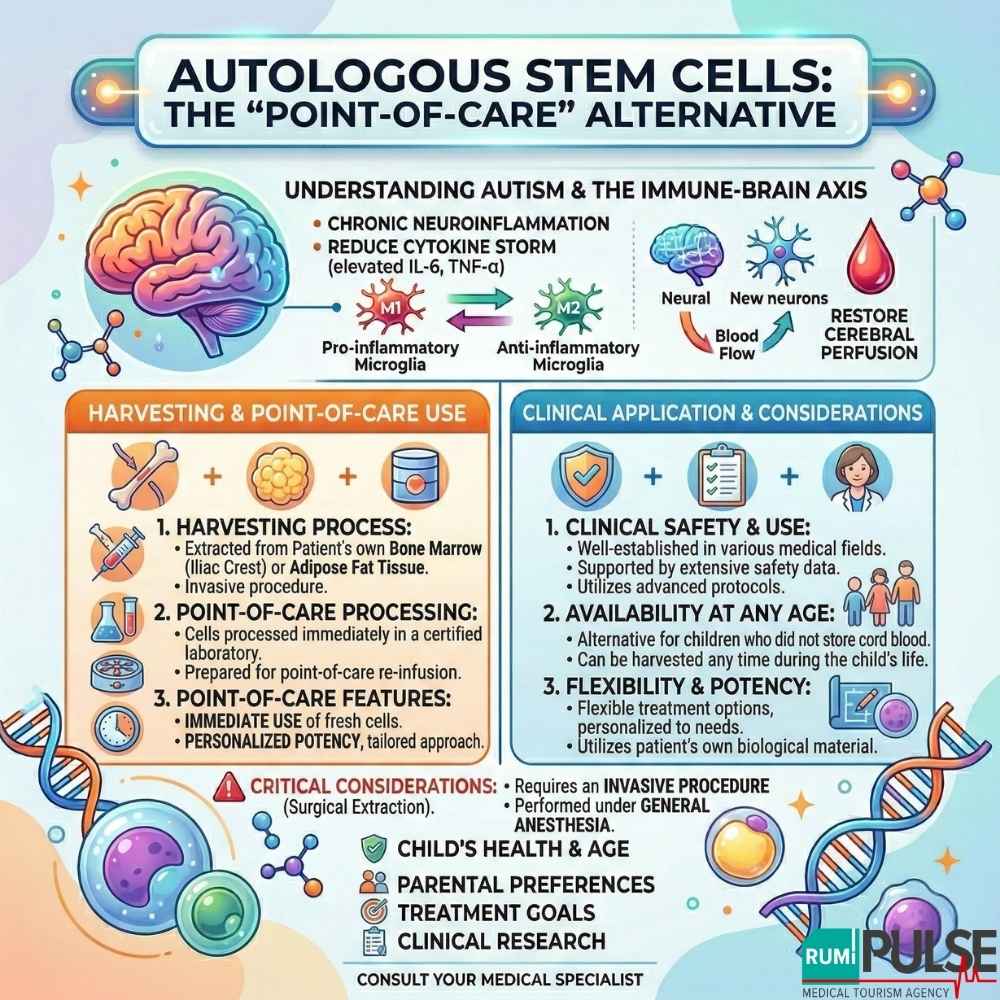
For families who did not store cord blood, Autologous Bone Marrow Mononuclear Cells (BMMNCs) offer a viable alternative using the child’s own biological material. These stem cells can be harvested from bone marrow or adipose tissue and are used immediately in a point-of-care therapy.
Autologous stem cell therapy is well-established in medical practice, with applications across multiple conditions. At Rumi Pulse, experts use advanced protocols to ensure the highest standards of cell viability and clinical outcomes. This therapy offers a personalized, flexible treatment option, particularly beneficial for children who did not store cord blood.
However, the primary drawback of autologous stem cells is the invasive procedure, as it requires general anesthesia and a surgical step to extract bone marrow. Additionally, some researchers argue that older children or those with high levels of systemic inflammation might experience lower stem cell quality, reducing the potential therapeutic effects.
For more on autologous stem cell therapy, check out Rumi Pulse Stem Cell Supported IVF.
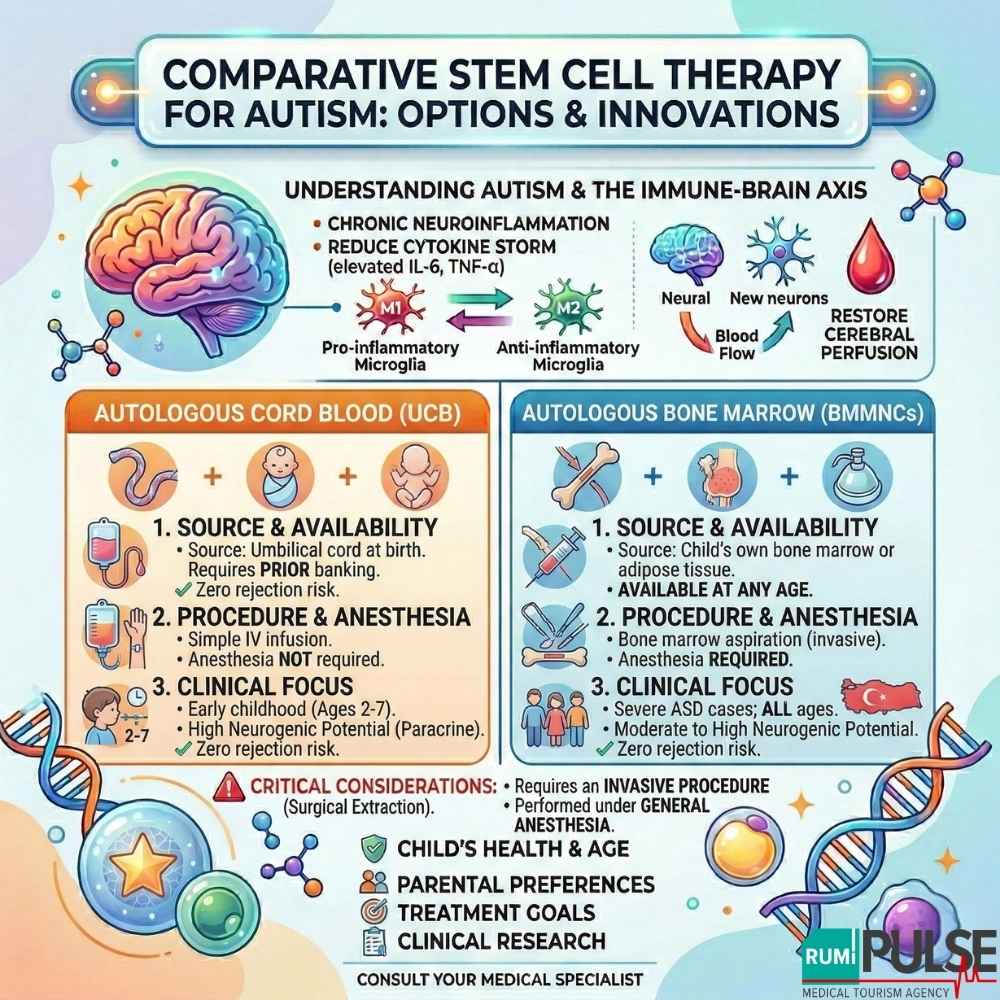
| Feature | Autologous Cord Blood (UCB) | Autologous Bone Marrow (BMMNCs) |
|---|---|---|
| Source | Umbilical cord at birth | Child’s own bone marrow or adipose tissue |
| Availability | Requires prior banking | Available at any age |
| Procedure | Simple IV infusion | Bone marrow aspiration (invasive) |
| Anesthesia | Not required | Required |
| Rejection Risk | None | None |
| Clinical Focus | Early childhood (Ages 2-7) | Severe ASD cases; all ages |
| Neurogenic Potential | High (Paracrine effects) | Moderate to High |
For children without stored cord blood, Wharton’s Jelly Mesenchymal Stem Cells (WJ-MSCs), derived from donor umbilical cord tissue, represent a growing alternative. These cells do not require HLA matching, meaning they can be used without fear of immune rejection. In many clinical centers in Turkey, WJ-MSCs are preferred due to their higher expansion capacity and potent anti-inflammatory effects.
The decision between cord blood and autologous stem cells depends on your child’s specific condition. Here is a decision matrix based on clinical data:
For a deeper understanding of your child's treatment options, schedule a consultation via our contact page.
Both cord blood stem cells and autologous stem cells offer promising potential for the treatment of autism, though they vary in availability, procedure, and benefits. The decision should be made after careful consideration of your child's specific needs, age, and the family's access to stored cord blood.
Consulting with a qualified expert in stem cell therapy can help ensure that you choose the best option for your child. To learn more about stem cell treatments and to get expert guidance, visit our contact page.

