

For children with Autism Spectrum Disorder (ASD), the search for treatments is often shaped by behavioral therapies and educational adjustments. However, regenerative medicine, including stem cell therapy, is adding a new dimension to this journey. Turkey has emerged as a global leader in stem cell therapy development, combining high-tech clinical innovations with strict safety standards, providing hope for families.
This post examines how stem cell therapy works for autism in Turkey, the scientific foundations behind it, and the treatment process in detail.
Turkey is not just a destination for medical tourism, but a center with a strong regulatory system. The Ministry of Health and the Turkish Medicines and Medical Devices Agency (TİTCK) oversee all treatment protocols, ensuring that each cell is processed in cGMP (Current Good Manufacturing Practice) certified laboratories. This system ensures that treatment processes are of international quality while prioritizing safety.
For detailed information about autism stem cell therapy, visit our Autism Therapy page or contact us via our Contact page.
Turkey is making significant scientific strides in autism treatment. Hacettepe University (PEDI-STEM) is at the forefront of pediatric stem cell research. Additionally, Ankara University is working on ethical and legal frameworks, ensuring that stem cell research in Turkey aligns with international standards.
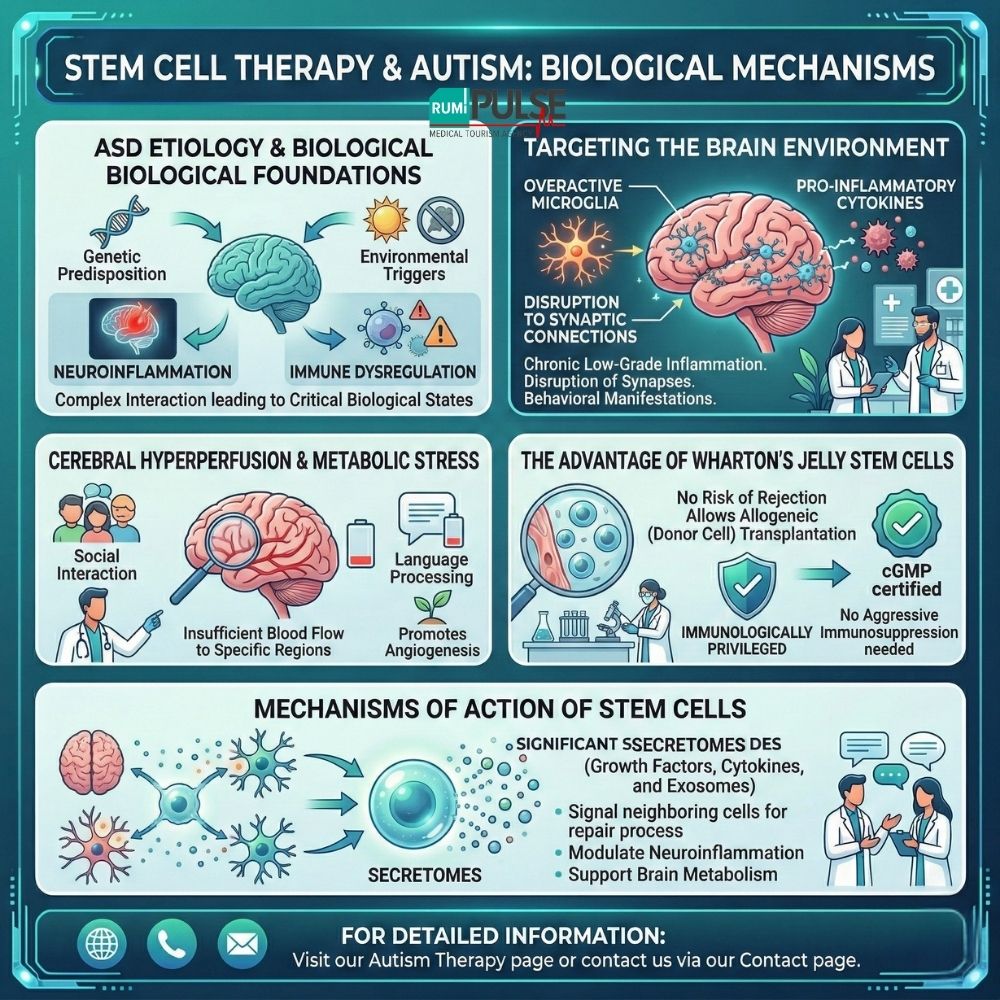
Autism Spectrum Disorder is a heterogeneous neurodevelopmental condition characterized by difficulties in social communication and the presence of repetitive behaviors. The etiology of autism is still an intense area of scientific research, but current findings suggest that it results from a complex interaction between genetic predisposition and environmental triggers. This interaction leads to two critical biological states: neuroinflammation and immune dysregulation.
Studies show that children with autism often have chronic low-grade inflammation in their central nervous system (CNS). The overproduction of molecules such as microglia and pro-inflammatory cytokines can lead to disruption in the brain’s normal synaptic connections, contributing to the behavioral manifestations of autism.
Another critical factor in children with autism is cerebral hypoperfusion, which means insufficient blood flow to specific regions of the brain. This condition negatively impacts metabolic activity, particularly in areas responsible for social interaction and language processing. Stem cell therapy aims to target this hypoperfusion by promoting angiogenesis—the formation of new blood vessels in the brain.
The most commonly used and researched stem cells in Turkey for autism therapy are derived from Wharton’s jelly (umbilical cord tissue). These cells occupy a unique developmental niche between embryonic and adult stem cells.
Wharton’s jelly cells are immunologically privileged, meaning they do not carry the risk of rejection by the immune system. This allows for allogeneic (donor cell) transplantation without the need for aggressive immunosuppressive treatments.
The main benefit of stem cells is often attributed to the release of “secretomes”—a cocktail of growth factors, cytokines, and exosomes that signal neighboring cells to begin the repair process. Scientific research shows that these mechanisms can help modulate neuroinflammation and support brain metabolism.
Stem cell therapy in Turkey is regulated through protocols set by the Ministry of Health. These protocols specify the source of the cells, the methods of administration, required diagnostic tests, and follow-up procedures. Turkey also sets an example globally with its high standards and ethical requirements for stem cell therapy.
In Turkey, stem cell therapies for neurological conditions must be approved by the Ministry of Health. This approval process ensures that the treatment is carried out professionally and ethically, and that clinics adhere to international standards. For clinic details and protocols, visit the Autism Therapy page.
Clinics in Turkey ensure that stem cells are pure, effective, and sterile by using cGMP-certified laboratories. These facilities are typically FDA-registered and adhere to international standards for sterile processing, ensuring the highest quality of cellular products.
For more clinic support, visit our Contact page.
The stem cell therapy process in Turkey is highly structured. Families go through an evaluation before treatment, the treatment itself, and post-treatment support.
A reputable clinic begins by remotely evaluating the child’s medical history. This initial step involves a multidisciplinary review by neurologists, pediatricians, and sometimes anesthesiologists. For more details, visit the Autism Therapy page.
Mesenchymal stem cells are administered through intravenous infusion or intrathecal injection. For most children, the treatment is minimally invasive and performed under professional supervision.
After the stem cell treatment, children are monitored for a few hours to ensure there are no adverse reactions. Many clinics in Turkey also offer adjunctive therapies such as Hyperbaric Oxygen Therapy (HBOT) or Glutathione Infusions to support cell survival and integration.

Clinical studies indicate that stem cell therapy for autism does not result in serious adverse effects. The most common observed effects are temporary and resolve quickly with symptomatic treatment. Here is a breakdown of possible side effects:
| Side Effect Type | Frequency | Duration / Management |
|---|---|---|
| Mild Fever | 10-15% | Resolves spontaneously within 24 hours or with antipyretic medication. |
| Injection Site Sensitivity | 5-10% | Lasts 1-2 days, can be managed with local ice application. |
| Headache (after I.T. injection) | 2-5% | Managed with lying position and fluid intake. |
| Immune Response | Very Rare | Typically non-pathogenic, mild rash. |
One of the main reasons families choose Turkey for stem cell therapy is its affordability compared to other countries. Below is a cost comparison:
| Country | Average Cost per Treatment Cycle (USD) | Inclusions |
|---|---|---|
| Turkey | $6,000 - $18,000 | Treatment, hotel, VIP transfers, translation |
| USA/UK | $25,000 - $50,000+ | Treatment, private trial access, logistics |
| Mexico | $12,000 - $25,000 | Treatment and basic support |
| Thailand | $15,000 - $30,000 | Treatment and some hospitality |
Data from clinics in Turkey shows that many children experience significant improvements within 3 to 6 months after treatment. These improvements are often seen in areas such as eye contact, behavioral stability, speech development, cognitive focus, and social interactions.

For personalized guidance or questions, visit our Contact page.
